Research program : Respiratory diseases and the environment
Home > Team 3 > Research program > Axe 1
Epigenetics and Chronic Lung Diseases: COPD and Cystic Fibrosis
Albertina De Sario, Arnaud Bourdin
Epigenetics and Chronic Lung Diseases: COPD and Cystic Fibrosis
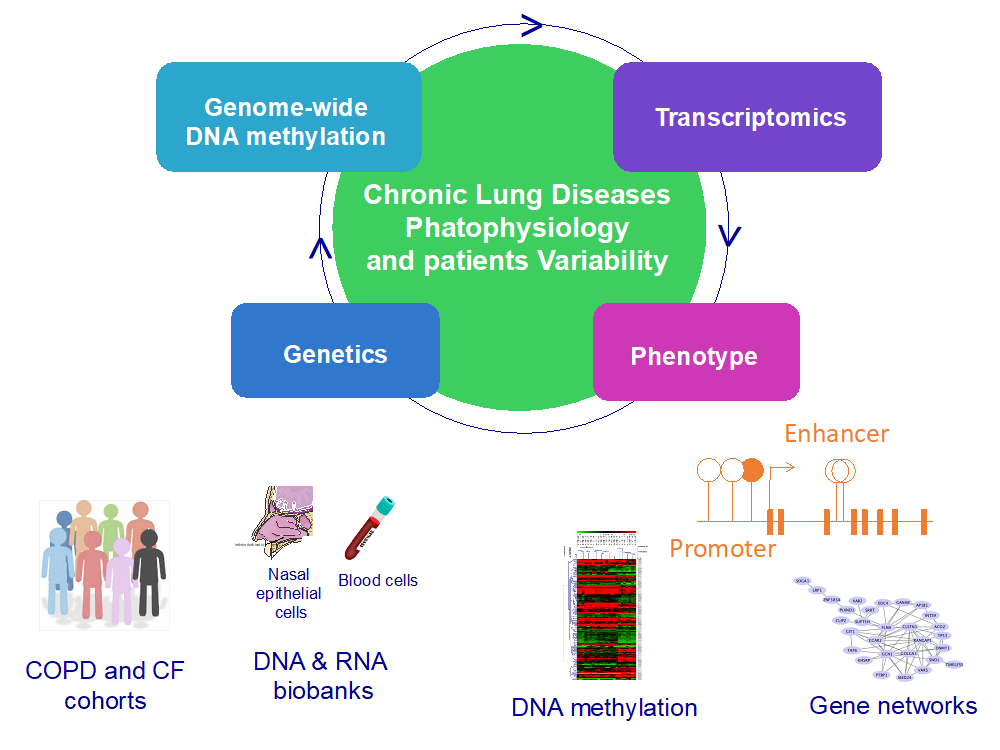
Epigenetic modifications regulate several biological processes, including gene transcription. They provide the cell with a fine and dynamic regulation of gene expression and allow the adaptation of organisms to the environment. An altered epigenetic regulation may either lead to chronic lung diseases or modulate their severity. Team n. 3 studies the role played by epigenetics in the etiology of chronic lung diseases and factors responsible for patient heterogeneity. Specifically, we address Chronic Obstructive Pulmonary Disease (COPD), a common disease, and Cystic Fibrosis (CF), a rare genetic disease.
Chronic Obstructive Pulmonary Disease (COPD) is characterized by an increased resistance to airflow in the small conducting airways, leading to progressive and incompletely reversible airflow obstruction and air trapping. Both a systemic inflammation and an abnormal inflammatory response in the lungs are observed, the latter one resulting in parenchymal destruction (emphysema). Multiple comorbidities are associated with COPD, namely asthma, lung cancer, diabetes and cardiovascular diseases. COPD is generally caused by exposure to noxious particles or gases (i.e. cigarette smoking, workplace pollutants, and/or biomass fuel smoke) with genetic factors also contributing to the disease. Cystic Fibrosis (CF) is a rare genetic disease that results from the impairment of CFTR (Cystic Fibrosis Transmembrane Conductance Regulator), a chloride channel responsible for ion transport across the plasmic membrane of epithelial cells. Pulmonary disease (chronic airway neutrophilic inflammation, recurrent infections and progressive lung function decline) is the first cause of morbidity and mortality in CF. Gastrointestinal manifestations and exocrine pancreatic insufficiency are also present and a number of comorbidities become more prevalent with age (i.e. diabetes and liver disease).
In both COPD and CF, the severity of the lung disease is variable and affected by genetic and environmental factors. In addition, variable inflammatory phenotypes (eosinophilia or neutrophilia) are observed among patients with COPD. A major effort is done by Team n. 3 to assess whether epigenetic factors account for the variable pulmonary and inflammatory phenotypes that are observed among patients with COPD and CF.
By combining epigenomics, genomics and bioinformatics, we assess whether DNA methylation accounts for the yet unexplained individual variability in the course of COPD and CF, searching genes and gene pathways responsible for the underlying inflammatory, cellular and molecular alterations. DNA methylation profiling is also implemented to disclose prognostic and predictive markers (response to new curative treatments).
Keywords: COPD, cystic fibrosis, DNA methylation, chromatin, biomarkers, on-chip hybridization, pyrosequencing, RNAseq, bioinformatics, cohorts, biobanks
Financements